There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. Please note that the elements do not show their natural relation towards each other as in the Periodic system. Number of electrons Number of protons Number of neutrons Number of isotopes Number of nucleons The atomic number is number of protons in the nucleus of an. The first chemical element is Hydrogen and the last is Ununoctium. 
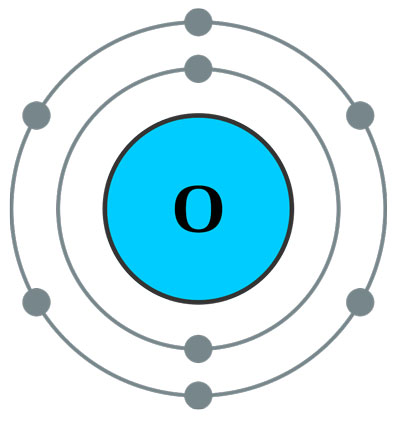
That means that all sodium atoms have 11 protons. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. In particular, it's the atom that links together the phosphate groups in the energy-carrying molecule ATP. Oxygen is also in virtually every molecule in your body including fats, carbohydrates and DNA. For example, the atomic number (z) for sodium (Na) is 11. Oxygen is a chemical element with atomic number 8 which means there are 8 protons and 8 electrons in the atomic structure. O, just about the most perfect solvent you can imagine for biochemistry. The symbol for the atomic number is designated with the letter Z. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. This number is known as the atomic number, which identifies the number of protons in the nucleus of ALL atoms in a given element. This list contains the 118 elements of chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
